Multiple Comorbidities Complicating Diagnosis of Polycythemia Vera
Presentation
J.J. is a 74-year-old male with a history of coronary artery disease for which he had a coronary artery bypass graft, and aortic stenosis for which he had a transcatheter aortic valve replacement (TAVR). He also has pulmonary hypertension and a pulmonary embolism (PE). He was referred to hematology for a hypercoagulable workup due to the recent PE and recommendations regarding the need for long-term anticoagulation.
J.J. states he tolerated the TAVR procedure well and there were no immediate complications. He was feeling energetic and had been taking walks daily. Three weeks post-procedure, he developed worsening fatigue and dyspnea with exertion. These symptoms prompted evaluation in the emergency department. A CT angiography of the chest revealed an acute pulmonary thromboembolism in his medial segmental/subsegmental right middle lobe pulmonary artery, and an enlarged main pulmonary artery, which may be suggestive of pulmonary hypertension. At that time, J.J. was diagnosed with a PE and started on anticoagulation with apixaban.
Labs
A review of prior laboratory studies revealed persistent leukocytosis and erythrocytosis with average hemoglobin levels of 17 g/dL and a white blood cell (WBC) count consistently > 11 x 10⁹/L. Peak hematocrit was 53.4% and peak WBC count was 15 x 10⁹/L. J.J. had repeat labs in the office at consultations, with the following results:
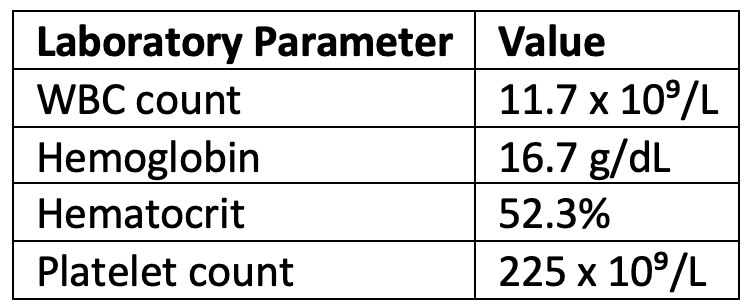
Workup
The presence of erythrocytosis and leukocytosis, especially in the setting of an unprovoked thrombosis, increases concern for possible underlying myeloproliferative neoplasm, namely polycythemia vera (PV).
The workup included a full hypercoagulable evaluation with a number of laboratory tests. Additionally, an erythropoietin (EPO) level and JAK2 mutation analysis were sent on the peripheral blood.
Results
The JAK2 V617F mutation returned positive and the EPO level was low-normal. This, together with J.J.'s clinical presentation, confirms a diagnosis of primary PV. He was set up for a bone marrow evaluation to determine a baseline, which was consistent with PV. J.J. was started on cytoreductive therapy, in addition to phlebotomy with a hematocrit goal of < 45%, to reduce his risk for another thrombotic event. He continued on apixaban and aspirin as other measures of thrombotic prevention.
Discussion
This patient presentation highlights multiple variables and the confounding effect that comorbidities can have on a myeloproliferative neoplasm diagnosis. The PE was considered to be unprovoked given that the TAVR was 3 weeks prior and J.J. was fully ambulatory post-procedure and doing well. His history of pulmonary hypertension could contribute to a secondary erythrocytosis and often inflammation can be the primary etiology for nonspecific leukocytosis. However, the presence of persistent leukocytosis and erythrocytosis warranted specific evaluation of a primary bone marrow disorder. Knowing that the JAK2 V617F mutation is present in 95% of patients with PV, it is an appropriate first step to order this test on the peripheral blood when this hematologic malignancy is on the differential.1 In this setting, there is no indication to obtain a full molecular mutational panel.
Reference
- Piris-Villaespesa M, et al. Prevalence of JAK2V617F in individuals that meet World Health Organization erythrocytosis criteria for polycythemia vera. Blood. 2018;132(suppl 1):1775.
