Patient With Polycythemia Vera and Severe Iron Deficiency Due to Frequent Phlebotomy
Presentation and Diagnosis
B.B. is a 66-year-old male who was diagnosed with JAK2V617F mutation–positive polycythemia vera (PV) in 2011. On presentation, his hematocrit (Hct) was 60%, white blood cell count 10.0 10*9/L, and platelets 510 10*9/L. Initially, he was in the low-risk category given his age < 60 and no prior thrombosis. He was started on low-dose aspirin and phlebotomized regularly to reach a target Hct of 45%. After 6 weeks of weekly phlebotomy, the frequency of therapeutic phlebotomy decreased to every 2 months and then every 3 months.
B.B. was somewhat lost to follow-up until last year when he presented due to increased symptoms of disease including significant pruritis (worse after a shower), fatigue, cognitive difficulties, and restless leg syndrome. He states that in the interim he was seen by a local provider closer to home and had a standing order for phlebotomies, which he would get as needed for Hct > 45%. Phlebotomy occurred on average every 3 months.
Additional Workup
Upon re-establishing care in our clinic, B.B.’s labs were obtained, and pertinent positives are as follows:
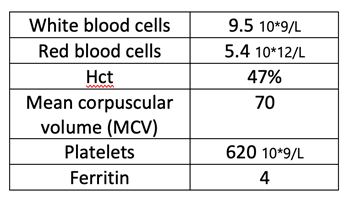
Risk Assessment/Treatment
At this point in time, B.B. is now considered high risk for a thrombotic event given that his age is > 60. Additionally, he needs frequent phlebotomy to maintain Hct < 45% and has uncontrolled symptoms of PV with the current approach to disease management. His restless leg syndrome and difficulty concentrating may be due to severe iron deficiency, which may have been present at disease onset but certainly exacerbated by many years of routine therapeutic phlebotomies. B.B. is advised to continue baby aspirin daily and start cytoreductive therapy with hydroxyurea. Phlebotomy may also need to continue until hydroxyurea has provided therapeutic benefit.
Interim Monitoring
B.B. returns for routine labs and follow-up every 2 weeks for the first month of treatment with hydroxyurea. More frequent follow-up is necessary after starting a new therapy to monitor closely for possible adverse events. He is tolerating hydroxyurea well and needs a phlebotomy 3 months post-initiation. His labs and evaluation visits are extended back to every 3 months, and at 6 months posttreatment initiation, his labs are significantly improved. B.B. is thankful that he does not need a phlebotomy as this is the first time in years he has not required therapeutic phlebotomy every 3 months.
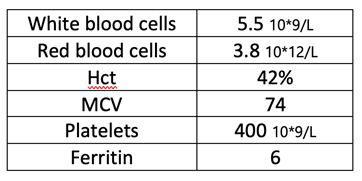
Considerations
Most patients with PV present with some degree of iron deficiency as evidenced by a low MCV, ferritin, and serum iron levels with an elevated transferrin and iron-building capacity. Additionally, thrombocytosis may worsen in the setting of iron deficiency. Iron deficiency is often exacerbated by therapeutic phlebotomy, as a desired effect of phlebotomy is thought to limit the rate of erythropoiesis by restricting this necessary part of hemoglobin synthesis. While phlebotomy does play an important role in controlling Hct levels to reduce risk of cardiovascular events, repeated phlebotomy may exacerbate iron deficiency–related symptoms such as pruritis, restless leg syndrome, and cognitive decline. However, the symptoms associated with iron deficiency may overlap with those of PV, which makes identifying the etiology somewhat difficult.
Current treatment practices do not include recommendations for treatment of iron deficiency, as iron supplementation will likely increase erythropoiesis, which is counterproductive in the PV setting. In rare cases of severe symptoms of iron deficiency, low-dose supplementation can be given with very close monitoring of Hct levels. An ideal treatment option would be to provide disease control in the absence of iron deficiency–related complications. Ruxolitinib as a second-line option may provide benefit in the setting of exacerbated symptoms with severe iron deficiency. In an exploratory analysis of the RESPONSE trial data, patients treated with ruxolitinib showed greater control of iron indices compared to best available therapy.1 Ruxolitinib may provide an opportunity to manage PV without dysregulation of iron homeostasis.
Reference
- Verstovsek S, et al. Markers of iron deficiency in patients with polycythemia vera receiving ruxolitinib or best available therapy. Leuk Res. 2017;56:52-59.
