A Patient With Myelofibrosis and Pancytopenia
Presentation
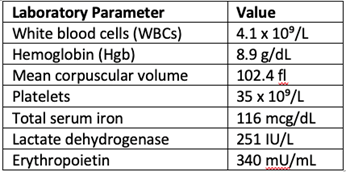
Mr. L is a 60-year-old male with past medical history significant for fatty liver disease who is initially referred to hematology for evaluation of thrombocytopenia with a platelet count of 80 x 10⁹/L. Workup includes upper abdominal ultrasound, which reveals hepatic steatosis and mild splenomegaly. A bone marrow biopsy is performed and identifies a hypercellular marrow (70%-80%) with patchy collagen fibrosis and grade 3/3 reticulin fibrosis and megakaryocytic atypia. There is no increase in blasts. Myelodysplastic syndrome FISH panel is negative, conventional cytogenetics are diploid, and JAK2 mutation is positive. Next-generation sequencing does not identify additional molecular mutations. Mr. L is diagnosed with primary myelofibrosis.
Laboratory studies at diagnosis:
Interval History
Mr. L is presented with options for treatment, including JAK inhibitor therapy with or without a hypomethylating agent. However, he opts for no therapy and pursues active surveillance.
Approximately 1 year later, he develops a right lower extremity deep vein thrombosis (DVT) and starts anticoagulation at a 50% dose due to severe thrombocytopenia. He continues to decline treatment for myelofibrosis.
Mr. L develops dependency to packed red blood cell transfusions, requiring 1 unit every month. He is started on erythropoietin stimulating agent weekly, which improves his hemoglobin levels slightly, yet he still requires frequent transfusions. He is referred for a second opinion, at which time a repeat bone marrow biopsy is performed.
Disease Restaging
Bone marrow biopsy results:
- Markedly hypercellular bone marrow (99%)
- Myeloid hyperplasia
- Megakaryocytic hyperplasia
- Megakaryocytic hyperplasia and dyspoiesis
- Grade 1 with focal grade 2/3 reticulin fibrosis
- JAK2+, no other molecular mutations
- Cytogenetics diploid
Prognostic Scoring
Mr. L’s Dynamic International Prognostic Scoring System Plus (DIPSS+) score is 4, which indicates high-risk disease. The DIPSS+ incorporates the following factors:
- Age <65
- Constitutional symptoms
- WBC >25
- Hgb <10
- Blasts >1% (2% in peripheral blasts)
- Diploid karyotype
- Transfusion dependency
- Platelets <100,000
Treatment
Given Mr. L’s good performance status and high-risk disease, he is again recommended for stem cell transplant, per National Comprehensive Cancer Network (NCCN) guidelines.1 However, due to lack of social support, he continues to decline. He starts pacritinib (JAK2/IRAK1/FLT3 inhibitor) at standard dosing 200 mg BID. He then develops a rash, which is thought possibly to be secondary to pacritinib, which is held and then resumed after rash resolution. After resuming, the rash recurs; pacritinib is again held and prednisone is prescribed. Mr. L’s rash resolves, and he restarts pacritinib at 50% dose reduction (100 mg BID) and continues to do well with excellent tolerance.
Discussion
Since starting pacritinib and taking it consistently, Mr. L’s packed red blood cell transfusion burden has decreased, although he still requires occasional transfusion support. His platelets have remained in the 30,000-50,000 range without the need for platelet transfusion, which is critical as he remains on anticoagulation given prior DVT. White blood cells remain low, yet he has not developed neutropenia or frequent infections.
Patients with myelofibrosis and cytopenias pose a therapeutic challenge. JAK1/2 inhibitors have myelosuppressive effects and thus can exacerbate transfusion dependency and the need for blood count support, especially in patients with cytopenias at baseline. Pacritinib, which does not significantly inhibit JAK1,2 is approved for patients with myelofibrosis and severe thrombocytopenia, defined as a platelet count less than 50 x 109/L. In addition to this designation, the NCCN has recommended pacritinib as a second-line treatment in low-risk and high-risk patients with myelofibrosis who have a platelet count greater than 50 x 109/L who are also not candidates for transplant.1
References
- NCCN Guidelines for Myeloproliferative Neoplasms V.3.2022. https://www.nccn.org/professionals/physician_gls/pdf/mpn.pdf
- Verstovsek S, et al. The oral JAK2/IRAK1 inhibitor pacritinib demonstrates spleen volume reduction in myelofibrosis patients independent of JAK2V617F allele burden. Blood(2019) 134 (Supplement_1): 1674.
