Skin Rash Post Allogeneic Stem Cell Transplant, Treatment of Acute Skin GVHD
Presentation
A 32-year-old female patient presented with a maculopapular erythematous itchy rash across portions of her chest, shoulders, face, scalp, and arms. She has an underlying history of acute myeloid leukemia and had received an allogeneic stem cell transplantation from a matched unrelated male donor; the rash presented on day 42 after the transplant. Both the patient and the donor were cytomegalovirus (CMV) infection positive and had O positive blood type.
Pre-Transplant Status
The patient’s pre-transplant hematopoietic cell transplantation–specific comorbidity index (HCT-CI) was 0, and she was MRD negative. Her pre-transplant conditioning regimen was busulfan and cyclophosphamide.
Workup
Following the transplant, GVHD prophylaxis consisted of post-transplant cyclophosphamide and tacrolimus dosed according to trough levels with a goal of 5-10 µg/mL and 1000 mg of mycophenolate mofetil (MMF) three times a day. The MMF was discontinued 35 days after the transplant and the patient had one episode of diarrhea and then presented with the rash.
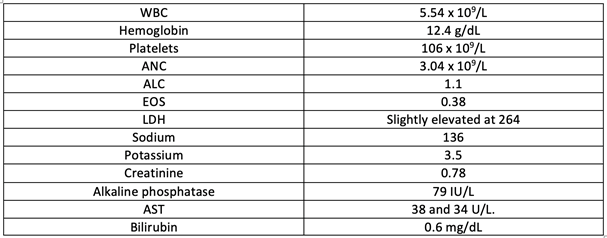
Neither CMV PCR nor EBV PCR was detected, and the STR post-transplant chimerism, the state in which the donor’s cells have durably engrafted to the patient, was greater than 98% without any recipient DNA detected. Standard lab values were as follows:
A symptomatic onset GvHD panel including both biomarkers ST2 and Reg3alpha was conducted. Results were as follows:

An acute GVHD (aGVHD) Symptomatic Algorithm analyzes the risk of severe aGVHD and non-relapse mortality (NRM), such as onset of GVHD symptoms and if the patient has not yet been treated, was also utilized. A score of greater than 0.290 means high risk of severe aGVHD and NRM and one of less than 0.140 indicates low risk of severe aGvHD and NRM. The patient’s score was 0.122.2
Assessment
The rash was evaluated and determined to be slightly less than 25% of body surface area (BSA) using the Wallace rule of nines. This classified the patient as having stage 1 aGVHD of the skin, without clear gastrointestinal or liver involvement. The overall GvHD grade was 1.1
Treatment
Topical steroid cream was given for the skin rash. For the body, the care team prescribed triamcinolone 0.1% three times daily as needed, and for the face, triamcinolone 0.025% cream for up to twice daily dosing. Labs were repeated, and an advanced practice provider evaluation was arranged for 4 days later.
Stool studies were ordered to screen for infectious diseases, but the patient was unable to provide a sample. She was given instructions to return to the clinic for a stool sample should her diarrhea continue or if it recurred. The patient was scheduled for repeat labs and a provider visit. In addition, a skin punch biopsy was scheduled in case the rash did not improve.
She presented back to the clinic 4 days later. Despite the topical steroid cream, the patient had a continued persistent and worsening of the skin rash that had now spread to more than 50% of her BSA. There was no bullous formation or desquamation or generalized erythroderma. Liver function tests remained within normal limits, and the patient had no additional loose stools. Overall, she had clinical grade II GVHD.1
The patient was also categorized as having stage 3 GvHD of the skin, and the skin punch biopsy was performed. It revealed superficial, perivascular infiltrate of lymphocytes with interface change and scattered dyskeratotic keratinocytes, overall being finalized as interface dermatitis consistent with GVHD.
Prednisone was administered at 1 mg/kg of the patient’s body weight, which was 70 mg a day, and she continued on therapeutic tacrolimus. Her progress was monitored, and as the rash did not improve following 2 weeks, the patient was deemed steroid-refractory.
Ruxolitinib was then started at 10 mg twice daily, and there was improvement in the skin findings.3 The erythematous rash resolved within approximately 28 days, leaving hyperpigmentation and dry skin but no additional erythema. Steroids were able to be tapered more quickly, and the patient remained on tacrolimus and ruxolitinib following discontinuation of steroids over the next 8 weeks.4
Discussion
Although GVHD is a clinical diagnosis, skin punch biopsy is helpful in determining the etiology of the rash. If diarrhea had persisted, the infectious workup would have included a number of stool studies including PCR testing of bacterial, viral, and parasites. If those remained negative, a colonoscopy for tissue that proved diagnosis of gut GVHD would have helped. In a severely immunocompromised patient after allogeneic stem cell transplantation, it is important to rule out other etiologies to minimize potential detrimental effects of adding high-dose steroids, as these are a mainstay of treatment of GVHD but may make infections worse. Close monitoring of viral testing in the blood, such as CMV PCR and EBV PCR, is important, as viral replication can occur, especially after the addition of steroids.
Topical steroids are preferred when total BSA is limited because steroids may also increase the risk of relapse of the patient’s underlying malignancy. Monitoring for relapse, as with MRD testing (PCR for applicable mutations, for instance) and STR post-transplant chimerism, should be performed regularly to monitor for any early signs of relapse post-transplant.
References
- Harris AC, Young R, Devine S, et al. International, multicenter standardization of acute graft-versus-host disease clinical data collection: a report from the Mount Sinai Acute GVHD International Consortium. Biol Blood Marrow Transplant. 2016;22(1):4-10. doi:10.1016/j.bbmt.2015.09.001
- Major-Monfried H, Renteria AS, Pawarode A, et al. MAGIC biomarkers predict long-term outcomes for steroid-resistant acute GVHD. Blood. 2018;131(25):2846-2855. doi:10.1182/blood-2018-01-822957
- Jagasia M, Perales MA, Schroeder MA, et al. Ruxolitinib for the treatment of steroid-refractory acute GVHD (REACH1): a multicenter, open-label phase 2 trial. Blood. 2020;135(20):1739-1749. doi:10.1182/blood.2020004823
- Mohty M, Holler E, Jagasia M, et al. Refractory acute graft-versus-host disease: a new working definition beyond corticosteroid refractoriness. Blood. 2020;136(17):1903-1906. doi:10.1182/blood.2020007336